Company Provides Clinical Data Update from Ongoing Phase 1 Clinical Trial with
ACTengine® IMA203 TCR-T Targeting PRAME
- Updated clinical data on ACTengine® IMA203 targeting PRAME in 30 heavily pre-treated metastatic melanoma patients at RP2D: 55% confirmed objective response rate, including tumor shrinkage achieved in 87% of patients; median duration of response of 13.5 months including 11/16 ongoing confirmed responses; IMA203 continues to maintain a favorable safety profile
- Registration-enabling randomized Phase 2/3 trial for ACTengine® IMA203 in 2L+ melanoma planned to commence in 2024 following further discussions with FDA
- Next data update on IMA203 and IMA203CD8 (GEN2) planned for 2H 2024
- First clinical data updates for Immatics’ next-generation, half-life extended TCR Bispecifics, TCER® IMA401 (MAGEA4/8) and TCER® IMA402 (PRAME), from ongoing Phase 1 dose escalation trials planned for 2H 2024; updates to include details on safety, pharmacokinetics and initial anti-tumor activity
$201.5 million public offering completed onJanuary 22, 2024 - Cash and cash equivalents, as well as other financial assets, amount to
$609.7 million 1 (€564.0 million) as ofMarch 31, 2024 funding company operations into 2027
“Our lead cell therapy candidate, IMA203, continues to show deep and durable responses in a significantly expanded data set since our last data readout in
First Quarter 2024 and Subsequent Company Progress
ACTengine® Cell Therapy Program
ACTengine® IMA203 monotherapy
Today,
As of the data cut-off on
- Confirmed objective response rate (cORR) of 55% (16/29)
- Disease control rate of 90% (27/30)
- Tumor shrinkage in 87% (26/30) of patients
- Median duration of response (mDOR) was 13.5 months (min 1.2+, max 21.5+ months) including 11 of 16 confirmed objective responses ongoing at data cut-off and longest duration of response ongoing at >21 months after infusion
- Confirmed response rates are similar across all melanoma subtypes (56% (9/16) in cutaneous melanoma; 54% (7/13) in other melanoma subtypes)
To date, IMA203 has maintained a favorable safety profile with no treatment-related grade 5 events in the safety population (N=65 patients across all dose levels and all tumor types).
Best overall response for IMA203 at RP2D in melanoma
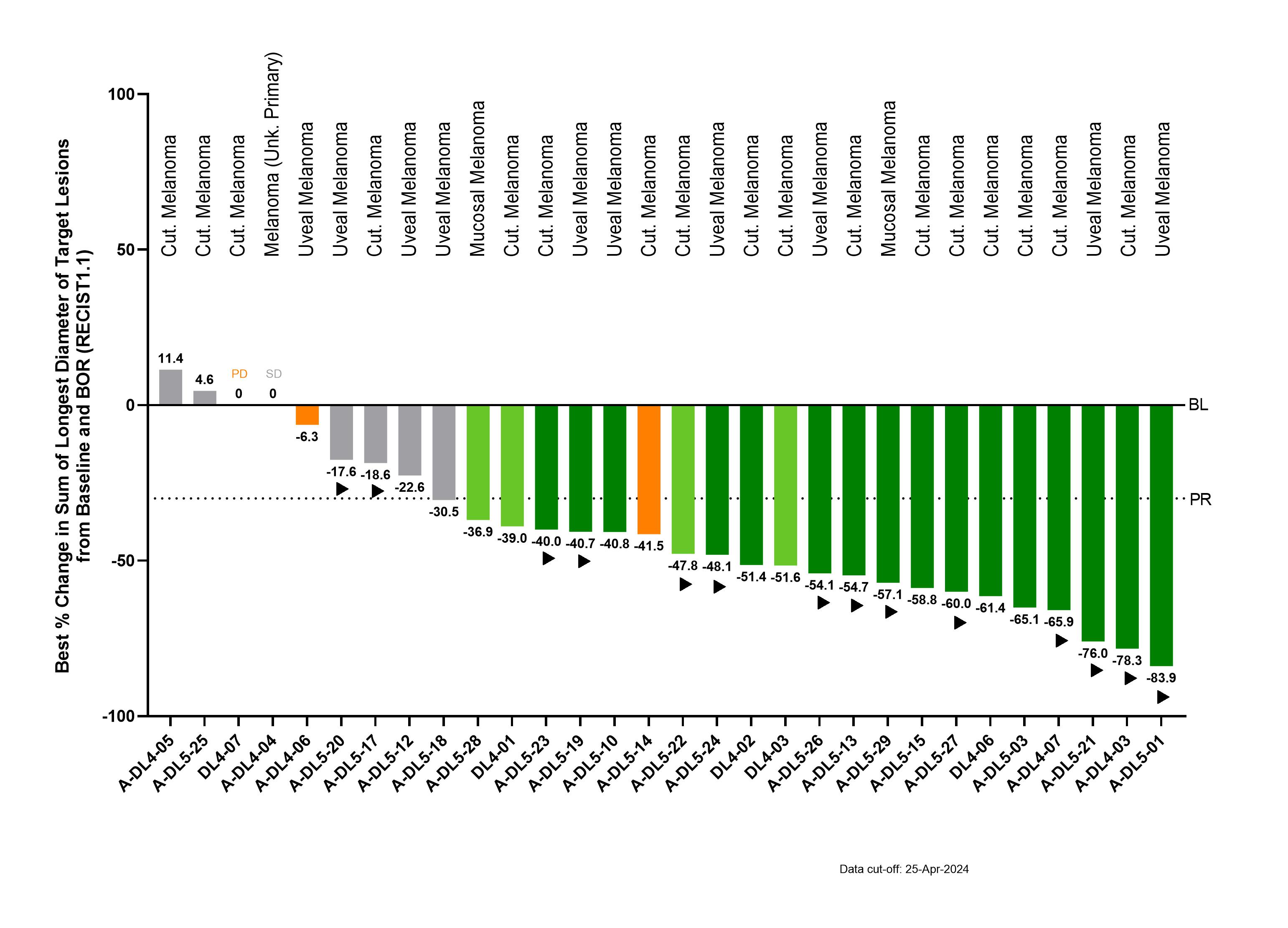
More information and details on the IMA203 clinical data update in melanoma are available in the
The next data update with translational and clinical data for IMA203 is planned for 2H 2024 at a medical conference.
Immatics’ late-stage clinical cell therapy development is supported by its differentiated manufacturing related to timeline, capabilities and facilities. ACTengine® IMA203 cell therapy products are manufactured within 7 days, followed by a 7-day QC release testing at a success rate of >95% to reach the target dose. The company has also recently completed construction of a ~100,000 square foot R&D and GMP manufacturing facility with a modular design for efficient and cost-effective scalability intended to serve early-stage and Phase 2/3 clinical trials, as well as initial commercial supply. The new site will start GMP manufacturing of cell therapy products in early 2025. Meanwhile, the existing GMP facility, which is run in collaboration with
Following an RMAT designation in
The Phase 2/3 trial is expected to assess IMA203 targeting PRAME in HLA-A*02:01-positive cutaneous melanoma patients versus a control arm. This approach is consistent with the FDA’s “one-trial” approach2, i.e., a single randomized controlled trial to support accelerated approval and the verification of clinical benefit to achieve full approval. The high prevalence of PRAME (≥95%) in cutaneous melanoma may enable the company to enroll patients without PRAME pre-testing. This would enhance trial operations and would remove the need to develop a companion diagnostic for PRAME testing in this indication. The full trial design is currently being developed and is subject to further alignment with the FDA as part of the ongoing discussions. Further details of the final clinical trial design will be provided in 2H 2024.
IMA203 is being developed to treat patients with metastatic melanoma, a prevalent cancer type with increasing incidence both inside and outside
ACTengine® IMA203CD8 (GEN2) monotherapy
As of the previously reported interim clinical update from
For IMA203CD8 (GEN2),
A next data update for IMA203CD8 (GEN2) is planned for 2H 2024.
Immatics’ T cell engaging receptor (TCER®) candidates are next-generation, half-life extended TCR Bispecific molecules. They are designed to achieve a patient-convenient dosing schedule and to maximize efficacy while minimizing toxicities in patients through the proprietary format using a high-affinity TCR domain against the tumor target and a low-affinity T cell recruiter binding to the T cell.
Upcoming milestones for Immatics’ clinical TCER® pipeline
Key objectives include:
- Demonstrating tolerability of the novel, next-generation, half-life extended TCR Bispecifics format;
- Optimizing dosing schedule to a less frequent regimen during dose escalation based on pharmacokinetics data;
- Demonstrating initial clinical anti-tumor activity (i.e., confirmed objective responses according to RECIST 1.1).
TCER® IMA401 (MAGEA4/8)
The Phase 1 dose escalation basket trial to evaluate safety, tolerability and initial anti-tumor activity of TCER® IMA401 in patients with recurrent and/or refractory solid tumors is ongoing. IMA401 targets an HLA-A*02:01-presented peptide that occurs identically in two different proteins, MAGEA4 and MAGEA8. This target peptide has been selected based on natural expression in native solid tumors at particularly high target density (peptide copy number per tumor cell identified by Immatics’ proprietary quantitative mass spectrometry engine XPRESIDENT® is >5x higher than for a MAGEA4 peptide target used in other clinical trials).
MAGEA4 and MAGEA8 are expressed in multiple solid cancers, including lung cancer, head and neck cancer, melanoma, ovarian cancer, sarcoma and others. Tolerability continues to be manageable with transient low-grade CRS, lymphopenia and neutropenia at high doses, all of which are expected for a bispecific T cell engager. A premedication with low doses of dexamethasone administered prior to the first 4 infusions, as used with other approved bispecific products, has been implemented as a preventative measure for continued dose escalation. Since the implementation of this premedication, to date, no cases of high-grade neutropenia among the patients treated have been observed. Based on pharmacokinetics data, the treatment schedule for IMA401 was switched from weekly to bi-weekly dosing. Confirmed objective responses have been observed in multiple patients.
IMA401 is being developed in collaboration with
TCER® IMA402 (PRAME)
Corporate Development
- On
January 22, 2024 ,Immatics completed an offering of 18,313,750 ordinary shares at a public offering price of$11.00 per share. The gross proceeds from the offering, before deducting the underwriting discount and offering expenses, were approximately$201.5 million .
First Quarter 2024 Financial Results
Cash Position: Cash and cash equivalents, as well as other financial assets, total €564.0 million (
Revenue: Total revenue, consisting of revenue from collaboration agreements, was €30.3 million (
Research and Development Expenses: R&D expenses were €32.1 million (
General and Administrative Expenses: G&A expenses were €11.6 million (
Net Profit and Loss: Net loss was €3.1 million (
Full financial statements can be found in the 6-K filed with the
Upcoming Investor Conferences
Bank of America Health Care Conference ,Las Vegas (NV) –May 14 - 16, 2024 Jefferies Global Healthcare Conference ,New York (NY) –June 5 - 7, 2024
To see the full list of events and presentations, visit www.investors.immatics.com/events-presentations.
- END -
About IMA203 and target PRAME
ACTengine® IMA203 T cells are directed against an HLA-A*02-presented peptide derived from preferentially expressed antigen in melanoma (PRAME), a protein frequently expressed in a large variety of solid cancers, thereby supporting the program’s potential to address a broad cancer patient population. Immatics’ PRAME peptide is present at a high copy number per tumor cell and is homogeneously and specifically expressed in tumor tissue. The peptide has been identified and characterized by Immatics’ proprietary mass spectrometry-based target discovery platform, XPRESIDENT®. Through its proprietary TCR discovery and engineering platform
ACTengine® IMA203 TCR-T is currently being evaluated in Phase 1 IMA203 monotherapy, and IMA203CD8 (GEN2) monotherapy, where IMA203 engineered T cells are co-transduced with a CD8αβ co-receptor. As previously reported, IMA203 in combination with an immune checkpoint inhibitor has been deprioritized.
About
Forward-Looking Statements
Certain statements in this press release may be considered forward-looking statements. Forward-looking statements generally relate to future events or the Company’s future financial or operating performance. For example, statements concerning timing of data read-outs for product candidates, the timing, outcome and design of clinical trials, the nature of clinical trials (including whether such clinical trials will be registration-enabling), the timing of IND or CTA filing for pre-clinical stage product candidates, estimated market opportunities of product candidates, the Company’s focus on partnerships to advance its strategy, and other metrics are forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “may”, “should”, “expect”, “plan”, “target”, “intend”, “will”, “estimate”, “anticipate”, “believe”, “predict”, “potential” or “continue”, or the negatives of these terms or variations of them or similar terminology. Such forward-looking statements are subject to risks, uncertainties, and other factors which could cause actual results to differ materially from those expressed or implied by such forward looking statements. These forward-looking statements are based upon estimates and assumptions that, while considered reasonable by
For more information, please contact:
| Media | |
| |
|
| Phone: +49 171 3512733 | |
| immatics@trophic.eu |
| |
|
| |
|
| Head of Strategy | |
| Phone: +1 346 319-3325 | |
| InvestorRelations@immatics.com |
Condensed Consolidated Statement of Loss of
| Three months ended March 31, |
||
| 2024 |
2023 |
|
| (Euros in thousands, except per share data) |
||
| Revenue from collaboration agreements | 30,269 | 9,796 |
| Research and development expenses | (32,108) | (27,581) |
| General and administrative expenses | (11,642) | (9,586) |
| Other income | 12 | 941 |
| Operating result | (13,469) | (26,430) |
| Change in fair value of liabilities for warrants | 1,043 | 7,397 |
| Other financial income | 11,381 | 2,795 |
| Other financial expenses | (677) | (3,509) |
| Financial result | 11,747 | 6,683 |
| Loss before taxes | (1,722) | (19,747) |
| Taxes on income | (1,332) | — |
| Net loss | (3,054) | (19,747) |
| Net loss per share: | ||
| Basic | (0.03) | (0.26) |
| Diluted | (0.04) | (0.26) |
Condensed Consolidated Statement of Comprehensive Loss of
| Three months ended March 31, |
||
| 2024 |
2023 |
|
| (Euros in thousands) | ||
| Net loss | (3,054) | (19,747) |
| Other comprehensive income | ||
| Items that may be reclassified subsequently to profit or loss | ||
| Currency translation differences from foreign operations | 336 | 564 |
| Total comprehensive loss for the year | (2,718) | (19,183) |
Condensed Consolidated Statement of Financial Position of
| As of |
||
| 2024 |
2023 |
|
| (Euros in thousands) | ||
| Assets | ||
| Current assets | ||
| Cash and cash equivalents | 122,093 | 218,472 |
| Other financial assets | 441,857 | 207,423 |
| Accounts receivables | 1,781 | 4,093 |
| Other current assets | 22,666 | 19,382 |
| Total current assets | 588,397 | 449,370 |
| Non-current assets | ||
| Property, plant and equipment | 49,968 | 43,747 |
| Intangible assets | 1,501 | 1,523 |
| Right-of-use assets | 11,886 | 13,308 |
| Other non-current assets | 1,373 | 2,017 |
| Total non-current assets | 64,728 | 60,595 |
| Total assets | 653,125 | 509,965 |
| Liabilities and shareholders’ equity | ||
| Current liabilities | ||
| Provisions | 1,740 | - |
| Accounts payables | 20,537 | 25,206 |
| Deferred revenue | 96,525 | 100,401 |
| Liabilities for warrants | 17,950 | 18,993 |
| Lease liabilities | 2,762 | 2,604 |
| Other current liabilities | 9,590 | 9,348 |
| Total current liabilities | 149,104 | 156,552 |
| Non-current liabilities | ||
| Deferred revenue | 91,358 | 115,527 |
| Lease liabilities | 11,877 | 12,798 |
| Other non-current liabilities | — | 4 |
| Total non-current liabilities | 103,235 | 128,329 |
| Shareholders’ equity | ||
| Share capital | 1,031 | 847 |
| Share premium | 1,001,402 | 823,166 |
| Accumulated deficit | (600,347) | (597,293) |
| Other reserves | (1,300) | (1,636) |
| Total shareholders’ equity | 400,768 | 225,084 |
| Total liabilities and shareholders’ equity | 653,125 | 509,965 |
Condensed Consolidated Statement of Cash Flows of
| Three months ended March 31, |
||
| 2024 |
2023 |
|
| (Euros in thousands) | ||
| Cash flows from operating activities | ||
| Net profit/(loss) | (3,054) | (19,747) |
| Taxes on income | 1,332 | — |
| Profit/(loss) before tax | (1,722) | (19,747) |
| Adjustments for: | ||
| Interest income | (6,294) | (2,254) |
| Depreciation and amortization | 3,014 | 1,811 |
| Interest expenses | 194 | 195 |
| Equity-settled share-based payment | 4,297 | 6,103 |
| Net foreign exchange differences and expected credit losses | (4,553) | 3,143 |
| Change in fair value of liabilities for warrants | (1,043) | (7,397) |
| Changes in: | ||
| Decrease/(increase) in accounts receivables | 2,312 | 880 |
| Decrease/(increase) in other assets | 574 | 234 |
| (Decrease)/increase in deferred revenue, accounts payables and other liabilities | (31,674) | (7,793) |
| Interest received | 2,484 | 1,189 |
| Interest paid | (194) | (79) |
| Income tax paid | — | — |
| Net cash (used in)/provided by operating activities | (32,605) | (23,715) |
| Cash flows from investing activities | ||
| Payments for property, plant and equipment | (9,174) | (4,317) |
| Payments for intangible assets | (2) | (8) |
| Proceeds from disposal of property, plant and equipment | — | — |
| Payments for investments classified in Other financial assets | (290,599) | (67,735) |
| Proceeds from maturity of investments classified in Other financial assets | 57,957 | 68,341 |
| — | — | |
| Net cash (used i n)/provided by investing activities | (241,818) | (3,719) |
| Cash flows from financing activities | ||
| Proceeds from issuance of shares to equity holders | 185,669 | — |
| Transaction costs deducted from equity | (11,548) | — |
| Payments related to lease liabilities | 524 | (866) |
| Net cash provided by/(used in) financing activities | 174,645 | (866) |
| Net (decrease)/increase in cash and cash equivalents | (99,778) | (28,300) |
| Cash and cash equivalents at beginning of the year | 218,472 | 148,519 |
| Effects of exchange rate changes and expected credit losses on cash and cash equivalents | 3,399 | (2,300) |
| Cash and cash equivalents at end of the year | 122,093 | 117,919 |
Condensed Consolidated Statement of Changes in Shareholders’ equity of
| (Euros in thousands) |
Share capital |
Share premium |
Accumulated deficit |
Other reserves |
Total share- holders’ equity |
| Balance as of |
767 | 714,177 | (500,299) | (1,481) | 213,164 |
| Other comprehensive income | — | — | — | 564 | 564 |
| Net loss | — | — | (19,747) | — | (19,747) |
| Comprehensive loss for the year | — | — | (19,747) | 564 | (19,183) |
| Equity-settled share-based compensation | — | 6,103 | — | — | 6,103 |
| Share options exercised | — | — | — | — | — |
| Issue of share capital – net of transaction costs | — | — | — | — | — |
| Balance as of |
767 | 720,280 | (520,046) | (917) | 200,084 |
| Balance as of |
847 | 823,166 | (597,293) | (1,636) | 225,084 |
| Other comprehensive income | — | — | — | 336 | 336 |
| Net loss | — | — | (3,054) | — | (3,054) |
| Comprehensive loss for the year | — | — | (3,054) | 336 | (2,718) |
| Equity-settled share-based compensation | — | 4,297 | — | — | 4,297 |
| Share options exercised | 1 | 682 | — | — | 683 |
| Issue of share capital – net of transaction costs | 183 | 173,257 | — | — | 173,440 |
| Balance as of |
1,031 | 1,001,402 | (600,347) | (1,300) | 400,786 |
1 All amounts translated using the exchange rate published by the
2 FDA Draft Guidance “Clinical Trial Considerations To Support Accelerated Approval of Oncology Therapeutics – Guidance for Industry,”
3 Estimated 41% HLA-A*02:01 positive population in the US; PRAME target prevalence is based on TCGA (for SCLC: in-house) RNAseq data combined with a proprietary mass spec-guided RNA expression threshold; Uveal melanoma target prevalence is based on IMADetect® qPCR testing of screening biopsies from clinical trial patients (n=33)
Attachment
 https://immatics.com/
https://immatics.com/


