Company to host conference call and webcast today,
- IMA402 and IMA401 TCR Bispecifics showed favorable tolerability at RP2D as well as deep and durable responses in heavily pre-treated, last-line patients with a range of solid tumors
- IMA402 PRAME Bispecific at RP2D range resulted in a 30% cORR (6/20) across all indications, including 29% (4/14) in melanoma and 2/3 confirmed responses in ovarian carcinoma
- IMA401 MAGEA4/8 Bispecific at ≥1 mg resulted in a 25% cORR (2/8) in head and neck cancer, 29% cORR (2/7) in melanoma and promising clinical activity in sqNSCLC
- Phase 1a dose escalation completed for both trials; data support IMA402 PRAME Bispecific development opportunities in cutaneous melanoma, gynecologic cancers and in combination with IMA401 MAGEA4/8 Bispecific in sqNSCLC
- Phase 1b dose expansion for IMA402 initiated
- Conference call and webcast can be accessed here
“Our off-the-shelf TCR Bispecifics have a proprietary next-generation format with half-life extension that is designed to combine optimized tolerability and potent anti-tumor activity while supporting patient-convenient dosing,” said
“Today marks the beginning of a new phase for
IMA402 PRAME Bispecific Phase 1a Dose Escalation Data Summary
Patient Population: Advanced metastatic solid tumors with no available treatment options
As of the data cutoff on
Safety: Treatment with IMA402 showed favorable tolerability
IMA402 showed favorable tolerability across a wide dose range in the 80 patients treated. The most frequent treatment-related adverse events (AEs) were expected and transient lymphopenia, consistent with the mechanism of action, and low-grade cytokine release syndrome (CRS): Grade 1: 33%, Grade 2: 5%, Grade 3: 0%, Grade 4: 1%. No ICANS or IMA402-related Grade 5 events occurred. Tolerability across all doses was consistent with tolerability at the RP2D range.
Phase 1a dose escalation in the monotherapy setting has been completed. The maximum tolerated dose (MTD) has not been reached. The provisional RP2D range has been identified at 10 to 30 mg. The Phase 1b dose expansion is ongoing at two distinct doses within the RP2D range, and the evaluation of IMA402 in combination with an immune checkpoint inhibitor has been initiated.
Anti-tumor Activity and Durability: Deep and durable responses observed at RP2D range
IMA402 showed a clear dose-response relationship across three different dose groups.
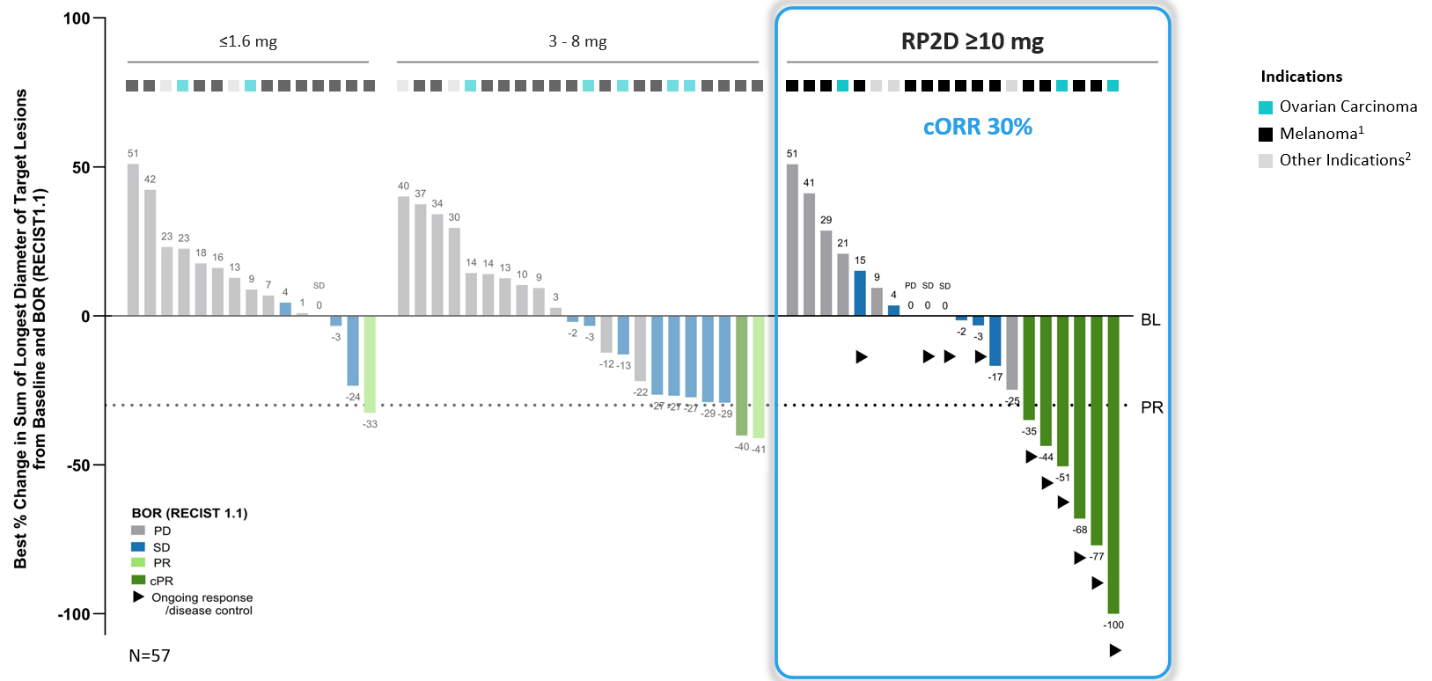
1 Melanoma includes cutaneous melanoma, melanoma of unknown primary, uveal melanoma; 2 Other indications include endometrioid carcinoma, synovial sarcoma and one patient with sqNSCLC at 1.6 mg; BL: baseline; BOR: best overall response; cORR: confirmed objective response rate; cPR: confirmed partial response; PD: progressive disease; PR: partial response; SD: stable disease; RECIST: response evaluation criteria in solid tumors; RP2D: recommended phase 2 dose
Several patients dosed with IMA402 at the RP2D range were observed to have deep and durable responses. All 6 confirmed objective responses were ongoing as of data cutoff, including two complete metabolic responses in cutaneous and uveal melanoma, ongoing at 8 and 18 months, respectively, as well as one confirmed partial response in ovarian carcinoma with -100% reduction in target lesions. All responders with ovarian carcinoma were platinum-resistant, and all responders with melanoma were immune checkpoint inhibitor-resistant.
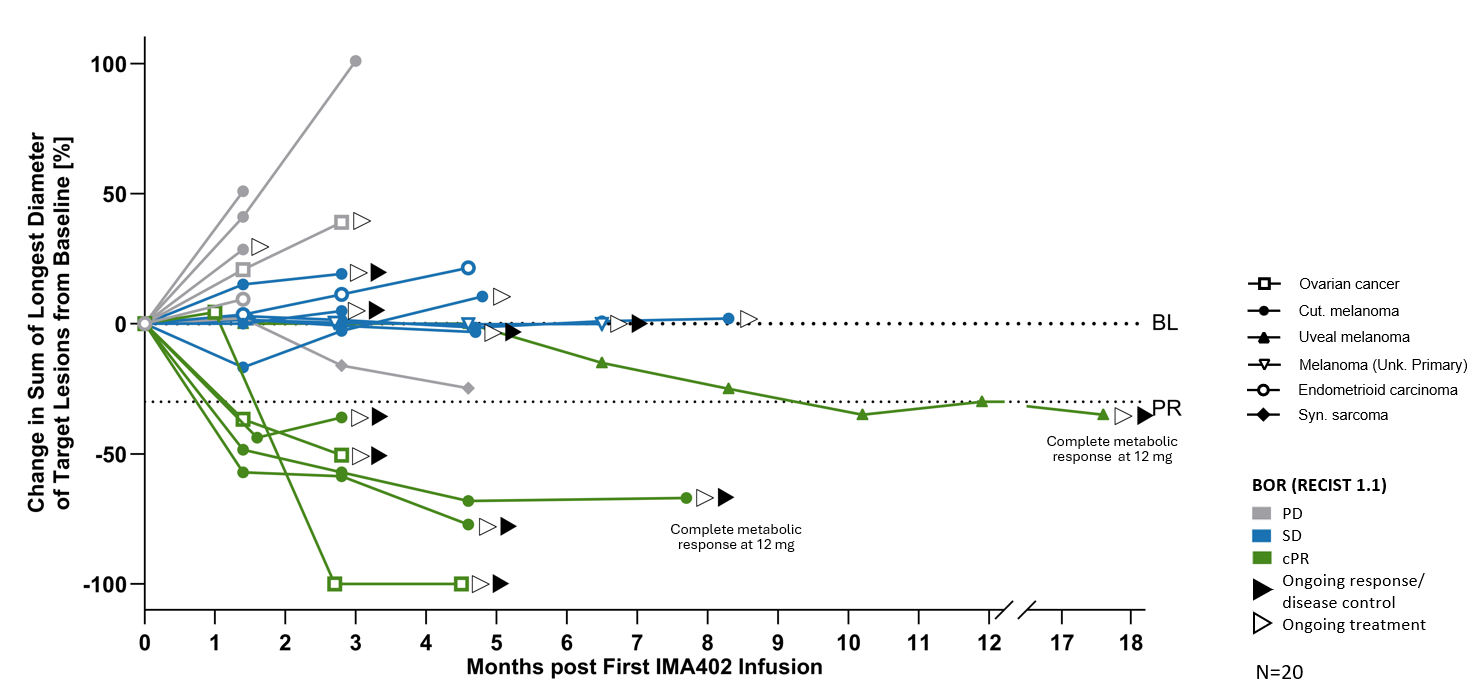
BL: baseline; cPR: confirmed partial response; PD: progressive disease; PR: partial response; SD: stable disease
Deep and durable responses at RP2D range (RECIST 1.1)
| All Indications |
Melanoma | Ovarian Carcinoma |
|
| cORR | 30% (6/20) | 29% (4/14) | 2/3 |
| mDOR, mFU month |
Not reached 4.2 |
Not reached 7.3 |
Not reached 2.2 |
| Tumor shrinkage | 55% (11/20) | 57% (8/14) | 2/3 |
| DCR (at week 6) | 65% (13/20) | 71% (10/14) | 2/3 |
mDOR: median duration of response; mFU: median follow-up; DCR: disease control rate
For patients across all indications treated within the RP2D range early, promising progression-free survival (PFS) and overall survival (OS) were observed:
- Median PFS was 4.8 months at a mFU of 6.8 months; 6-month PFS rate was 45%
- Median iPFS4 was not reached at a mFU of 6.3 months; 6-month iPFS rate was 58%
- Median OS was not reached at a mFU of 5.4 months; 1-year OS rate was 94%
Clinical Development Opportunities for IMA402 PRAME Bispecific
Based on the promising Phase 1a dose escalation data,
IMA401 MAGEA4/8 Bispecific Phase 1a Data Summary
Patient Population: Heavily pre-treated patients with a broad range of tumor types with no available treatment options
As of the data cutoff on
Safety: Treatment with IMA401 showed favorable tolerability at RP2D
The most frequent and relevant treatment-related adverse events (AEs) across all 55 patients treated with IMA401 were low-grade cytokine release syndrome (CRS) (24% G1, 11% G2, no ≥ Grade 3), mostly at the first step dose, expected and transient lymphopenia, consistent with the mechanism of action, as well as neutropenia, which was mostly transient, not re-occurring after resolution under continued treatment and well-manageable at the RP2D range of 1-2 mg. Notably, no ICANS was observed. The tolerability of IMA401 in combination with pembrolizumab is consistent with the tolerability of IMA401 monotherapy.
The maximum tolerated dose (MTD) has not been reached; three dose-limiting events were observed at 2.5 mg. The Phase 1a dose escalation has been completed, and the provisional RP2D range has been identified at 1-2 mg. At RP2D, the tolerability profile was favorable.
Anti-tumor Activity and Durability: Promising clinical activity and deep and durable responses were observed in patients with head and neck cancer, melanoma and lung cancer treated at ≥1 mg
Patients in three focus indications treated with ≥1 mg of IMA401 as a monotherapy or in combination with pembrolizumab demonstrated clinical activity:
- Head and neck cancer: cORR of 25% (2/8), disease control rate of 63% (5/8)
- Melanoma: cORR of 29% (2/7), disease control rate of 57% (4/7)
- Squamous non-small-cell lung cancer: 1 partial response at first scan for a heavily pre-treated, ICI-resistant patient, 1 patient with stable disease for >4 months and overall survival of approximately 16 months, 1 patient with progressive disease with shrinkage of liver target lesions
The duration of all confirmed responses was longer than 6 months post treatment, with the longest response ongoing over 2 years in a patient with advanced cutaneous melanoma.
Clinical Development Opportunity for IMA401 MAGEA4/8 Bispecific
Consistent with Immatics’ focus on advancing its PRAME franchise, the Company is exploring IMA401 in combination with IMA402, starting with squamous non-small cell lung cancer (sqNSCLC). Based on the clinical proof-of-concept of both bispecific candidates, including the initial promising activity of IMA401 in head and neck cancer and sqNSCLC, as well as preclinical proof-of-concept data,
About Immatics TCR Bispecifics (TCER®)
Immatics’ next-generation half-life extended TCER® molecules are antibody-like “off-the-shelf” biologics that leverage the body’s immune system by redirecting and activating T cells towards cancer cells expressing a specific tumor target. The design of the TCER® molecules enables the activation of any T cell in the body to attack the tumor, regardless of the T cells’ intrinsic specificity. Immatics’ proprietary biologics are engineered with two binding regions: a TCR domain and a T cell recruiter domain. The TCER® format is designed to maximize efficacy while minimizing toxicities in patients. It contains a high-affinity TCR domain that is designed to bind specifically to the cancer target peptide on the cell surface presented by an HLA molecule. The antibody-derived, low-affinity T cell recruiter domain is directed against the TCR/CD3 complex and recruits a patient’s T cells to the tumor to attack the cancer cells. With a low-affinity recruiter aiming for optimized biodistribution and enrichment of the molecule at the tumor site instead of the periphery, TCER® are engineered to reduce the occurrence of immune-related adverse events, such as cytokine release syndrome. In addition, the TCER® format consists of an Fc-part conferring half-life extension, stability, and manufacturability. TCER® are “off-the-shelf” biologics and thus immediately available for patient treatment. They can be distributed through standard pharmaceutical supply chains and provide the opportunity to reach a large patient population without the need for specialized medical centers.
About PRAME
PRAME is a target expressed in more than 50 cancers.
About IMA402 PRAME Bispecific
IMA402 is a molecule from Immatics’ TCR Bispecifics (TCER®) pipeline directed against an HLA-A*02:01-presented peptide derived from PRAME
IMA402 is currently being evaluated in a
About IMA401 MAGEA4/8 Bispecific
IMA401 is a molecule from Immatics’ TCR Bispecifics pipeline that targets an HLA-A*02:01-presented peptide derived from two different cancer-associated proteins, melanoma-associated antigen 4 and/or 8 (“MAGEA4/8”). The MAGEA4/8 peptide has been identified and validated by Immatics’ proprietary mass spectrometry-based target discovery platform XPRESIDENT® and is presented at a 5-fold higher target density (copy number per tumor cell) than the MAGEA4 peptide targeted in other clinical trials.
IMA401 is currently being evaluated in a
About
Forward-Looking Statements
Certain statements in this press release may be considered forward-looking statements. Forward-looking statements generally relate to future events or the Company’s future financial or operating performance. For example, statements concerning timing of data read-outs for product candidates, observations from the Company’s clinical trials, the timing, outcome and design of clinical trials, the nature of clinical trials (including whether such clinical trials will be registration-enabling), the timing of IND, CTA or BLA filings, estimated market opportunities of product candidates, the Company’s focus on partnerships to advance its strategy, and other metrics are forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “may”, “should”, “expect”, “plan”, “target”, “intend”, “will”, “estimate”, “anticipate”, “believe”, “predict”, “potential” or “continue”, or the negatives of these terms or variations of them or similar terminology. Such forward-looking statements are subject to risks, uncertainties, and other factors which could cause actual results to differ materially from those expressed or implied by such forward-looking statements. These forward-looking statements are based upon estimates and assumptions that, while considered reasonable by
For more information, please contact:
Media
Phone: +49 151 74416179
immatics@trophic.eu
Head of Strategy
Phone: +1 346 319-3325
InvestorRelations@immatics.com
- END -
1 Cutaneous melanoma, uveal melanoma, synovial sarcoma, endometrial carcinoma, ovarian cancer, squamous non-small cell lung cancer.
2 Efficacy-evaluable patients: All patients treated as of
3 N=2 endometrioid carcinoma, n=1 synovial sarcoma.
4 iRECIST, developed by the
5 Basket trial with >15 different tumor indications.
6 Efficacy-evaluable patients: All patients treated as of
Attachment
 https://immatics.com/
https://immatics.com/


